Originally published : Tue, March 22, 2022 @ 9:35 PM
Updated : Tue, March 21, 2023 @ 3:46 PM
This is the second blog in a two-part series exploring how oligonucleotide modifications can support the advancement of nucleic acid therapeutics.
In our first post of the series, we introduced the challenges facing manufacturers of nucleic acid therapeutics. In this post, we will explore the technologies and chemical modifications available that can be harnessed to overcome the challenges of using nucleic acids for therapeutic applications.
Chemical modifications enable therapeutic applications
Nuclease-resistant modifications
Nuclease resistance is essential for the therapeutic efficacy of nucleic acids. Nuclease-resistant modifications can be introduced to reduce or eliminate an oligonucleotide’s susceptibility to in vivo enzymatic degradation. Common modifications to enhance the nuclease resistance of antisense oligonucleotides (ASOs) and short interfering RNA (siRNA) oligonucleotides include phosphorothioate linkages and 2′-OMethyl (2′-OMe) and 2′-Fluoro (2′-F) bases. Reverse amidites also provide protection against enzymatic degradation.
Phosphorothioate bonds stabilise the oligonucleotide backbone against nuclease degradation, effectively enhancing its half-life in the cell. Oligonucleotides with phosphorothioate bonds can silence gene expression by directly blocking translation (a steric mechanism) or by enabling enzymatic degradation of the target transcript with an RNase H-mediated mechanism.
Substituting 2′-OMe bases at some or all positions of an oligonucleotide is a more common approach to induce nuclease resistance. Compared with phosphorothioate oligonucleotides, 2′-OMe modifications deliver higher nuclease resistance with reduced toxicity.1 However, the ability to induce RNase H cleavage of the targeted RNA may be limited unless gapmers are used in the oligonucleotide design.
Backbone modifications
|
Backbone modifications can increase the stability, binding affinity and nuclease resistance of a therapeutic oligonucleotide. Common backbone modifications include peptide nucleic acids (PNA) and locked nucleic acids (LNA). PNAs are oligonucleotide analogues in which a pseudo-peptide backbone substitutes the standard sugar-phosphate DNA backbone. The lack of phosphate in their peptide backbone grants PNAs a neutral charge, high binding affinity, chemical stability and greater selectivity than a standard unmodified oligonucleotide. |
Discover the
|
LNAs are a class of high-affinity RNA analogues that uses a methylene bridge to effectively “lock” the molecule’s ribose ring in an ideal conformation for Watson-Crick binding. The locked structure confers desirable features for therapeutic applications, including nuclease resistance, excellent target specificity and binding and increased thermal stability. LNA-based therapeutic molecules have the potential to potently and specifically inhibit RNA targets across a variety of tissues, making them a valuable tool for drug discovery and development.
Cell delivery and uptake modifications
In addition to stabilising RNA constructs in vivo, efficacious nucleic acid therapeutics require efficient, targeted delivery to the appropriate tissues. Optimised cell delivery platforms can help reduce dosage requirements, increase efficiency and minimise toxicity with more accurate tissue targeting. Typical cell delivery and uptake technologies include lipophilic modifications, vitamin based modifications and N-acetylgalactosamine (GalNAc) conjugates.
Lipophilic modifications decrease the solubility of an oligonucleotide in vivo so that it can reach target sites, including membranes, lipids and hydrophobic proteins. Lipophilic modifications, including cholesterol and palmitic acid, also facilitate passage through lipid membranes for improved cell penetration and tissue uptake.
Efficient, targeted drug delivery to the liver is possible with a carbohydrate called GalNAc. The molecule facilitates oligonucleotide delivery to liver hepatocytes by targeting the asialoglycoprotein receptor (ASGPR), found almost exclusively in liver cells. GalNAc conjugates enhance RNAi hepatocyte delivery and potency, resulting in lower doses or less frequent dosing.2
Phosphonate modification
In siRNA applications, 5′-phosphate is required for loading into RNA-induced silencing complex (RISC). Unfortunately, phosphorylation can be challenging because of the extensive chemical modifications required for in vivo stability of the siRNA molecule. Adding a synthetic 5′-Vinylphosphonate modification to conjugated siRNAs enhances stability, protects the molecule from phosphatases and improves silencing activity.3 Research shows that a 5′-Vinylphosphonate modification can improve siRNA accumulation and residence time in targeted tissues and enhance in vivo siRNA potency by improving Argonaute-2 binding.4
Solid supports for nucleic acid therapeutics
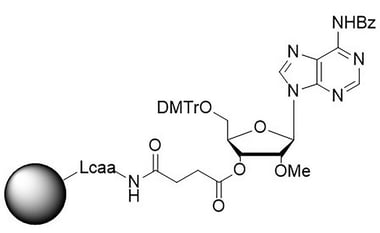 rA (Bz), 2′-OMe CNA CPG for the incorporation of a 2′-O-methyl modified ribo-A nucleobase at the 3′ end of an oligonucleotide.
rA (Bz), 2′-OMe CNA CPG for the incorporation of a 2′-O-methyl modified ribo-A nucleobase at the 3′ end of an oligonucleotide.
Solid support performance can be a significant factor in reducing oligonucleotide synthesis cost and ensuring a high-quality product. Physical parameters of the solid support, including particle size and shape, pore size, pore volume and surface area can affect the oligonucleotide’s purity and yield. Controlled pore glass (CPG) products specifically designed for the 2′OMe and 2′F modifications used in nucleic acid therapeutics applications offer the specifications needed to obtain a high-quality product for commercial applications.
Promising technology with a bright future
Nucleic acid therapeutics are a promising technology for managing debilitating diseases. The discovery and implementation of chemical modifications to improve an oligonucleotide’s stability and cell delivery can help overcome some of the challenges that prevent widespread clinical use.
LGC, Biosearch Technologies is proud to support the effort to bring life-changing drugs to the patients that need them most. Our extensive product portfolio includes modifications that are critical to optimising the efficacy of therapeutic applications. A long history of research and development supporting therapeutic applications, combined with our technical expertise, enables us to tackle complex drug development challenges with a toolbox of innovative solutions. In fact, more than 100 nucleic acid drugs, approved or in development, are made using at least one of our reagents. With convenient online ordering, reliable technical support and industry-leading products, Biosearch Technologies simplifies the path from drug discovery to commercialisation.
Explore our toolbox for oligonucleotide therapeutic development

|
Feeling ready to synthesise your own oligos?There are undeniable benefits to making your own oligos. However, beyond instrument selection there are a number of other considerations to work through such as solvent storage, disposal, ventilation, and other practicalities to ensure that your synthesizers run at full capacity and that you optimise your return on investment. Download a copy of this guide to support your decision making and a plan of action. |
References
- Kurreck J. The role of backbone modifications in oligonucleotide-based strategies. Therapeutic Oligonucleotides. Chapter 1:1-18. Published 2008. Accessed 3 September 2021. https://doi.org/10.1039/9781847558275-00001.
- Springer AD, Dowdy SF. GalNAc-siRNA Conjugates: Leading the Way for Delivery of RNAi Therapeutics. Nucleic Acid Ther. 28(3):109-118. Published 2018. Accessed 7 March 2022. https://doi.org/10.1089/nat.2018.0736.
- Haraszti RA, Roux L, Coles AH, et al. 5΄-Vinylphosphonate improves tissue accumulation and efficacy of conjugated siRNAs in vivo. Nucleic Acids Res. 45(13):7581-7592. Published 2017. Accessed 28 February 2022. https://doi.org/10.1093/nar/gkx507.
- Hu B. Zhong L, Weng Y, et al. Therapeutic siRNA: state of the art. Sig Transduct Target Ther. 5(101). Published 2020. Accessed 28 February 2022. https://doi.org/10.1038/s41392-020-0207-x.



