Originally published : Thu, August 4, 2022 @ 10:47 PM
Updated : Tue, August 9, 2022 @ 7:10 PM
New approaches that use aptamers as targeting agents may open the door to wider clinical use in the future
Despite their many potential benefits as therapeutics – namely a high thermal stability, the ability to bind tightly to virtually any target, easy and scalable production, and batch-to-batch uniformity1 – aptamers have seen little success in the clinic.2 In addition to rapid renal clearance and susceptibility to nucleases, aptamer performance in vitro against purified components has not always translated to similar performance in vivo.2
But recent efforts to harness aptamers as the targeting component of a larger therapeutic may finally see these promising molecules reach their full potential in the clinic. In this blog post we highlight a few recent applications of aptamers in therapeutic development, and look forward to seeing these innovations reach the market and advance human health in the near future.
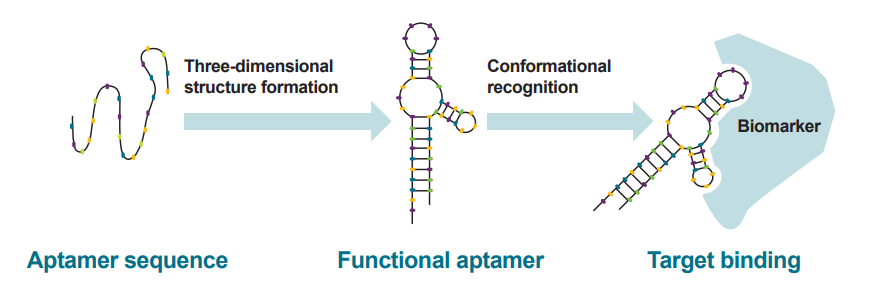
Figure 1. Nucleic acid aptamers are single stranded oligonucleotides (DNA or RNA), which fold into well defined three-dimensional structures, forming shapes with complementary interactions with a desired target (e.g. small molecules, proteins, cells and even whole organisms).3,4
Aptamers plus nanoparticles are delivering photodynamic therapies in cultured cells and small animal models
Photodynamic and photothermal therapies (PDT and PTT, respectively) are promising approaches for treating cancer,5 and are already used for a few indications today.6,7 When it works, PDT can be effective at killing tumour cells by inducing apoptosis. However, PDT is often challenged by its own success. Many of the first generation approaches use photosensitising agents that generate singlet oxygen, 1O2, which creates hypoxic conditions within the tumour microenvironment (TME). While there may be an initial apoptotic response that kills a few tumour cells, the hypoxic conditions activate protective signaling cascades that promote vascularisation and tumour growth, overcoming the PDT.
Newer approaches like PTT try to overcome the hypoxia challenge by using photosensitising agents that generate heat rather than singlet oxygen.
Regardless of the modality of tumour cell killing, a number of teams are looking to make both PDT and PTT safer by adding targeting signals that localise the sensitising agent to the tumour site. By combining a photosensitising agent with a tumour cell-specific aptamer on a nanoparticle scaffold, researchers are creating targeted PDTs and PTTs that are showing promising activity in cell and animal models.8–10
Aptamers on nanodroplets and lipid nanoparticles are delivering siRNA therapeutics
Another promising therapeutic approach that has been slow to reach commercialisation are small RNA-based medicines like siRNAs and miRNAs. The obvious challenge with small RNAs is prolonging their persistence in the bloodstream long enough to have a therapeutic effect. Nanomaterials including lipid nanoparticles and nanodroplets are currently being explored as delivery vehicles, and have already been shown to be spectacularly effective in their use to deliver COVID-19 mRNA vaccines.11
|
Here, aptamers are again coming into play, acting as selective moieties for targeted delivery. Because Guo, et al.12 were already using the nucleic acid-based phi29 DNA packaging motor (3WJ-pRNA) and delivering an siRNA as the therapeutic, adding an RNA aptamer for targeting simplified construction of their therapeutic. They were able to create a therapeutic molecule based completely on RNA – the active siRNA, targeting aptamer, and 3WJ-pRNA scaffold – that could then be anchored to a nanodroplet for stability. In another example, aptamers specific for the SARS-CoV-2 spike protein’s receptor binding domain (RBD) were used to target a lipid nanoparticle containing RNAi against the SARS-CoV-2 nucleocapsid phosphoprotein.13 The authors administered this therapeutic through a nebuliser for six days to a 36-year-old patient with severe COVID-19 and were able to show improvement in lung structure in CT scans. |
|
Aptamers are also being directly added to therapeutic molecules
One last application of aptamers to target therapeutics is their conjugation directly to the active therapeutic. Several recent examples include aptamers being added to siRNAs for oncological applications14 as well as aptamers added to thrombin for anticoagulant applications.15
An exciting future for aptamers
With a flurry of papers covering aptamers for use in targeting therapeutics, as well as a few more exploring the use of aptamers as therapeutics, the team at LGC Biosearch Technologies is looking forward to seeing aptamers in commercialised therapeutics in the not-too-distant future.
Biosearch Technologies’ can support your aptamer innovations with our extensive product portfolio of oligonucleotide modifications, solid supports and reagents. These include modifications for stability and nuclease resistance – such as locked nucleic acids and 2′-F and 2′-OMe phosphoramidites. We also supply 2′-F and 2′-OMe solid supports and reagents for phosphorothioate linkages. Explore the toolbox for oligonucleotide therapeutics to discover more.
Looking for a partner to manufacture your aptamer? Learn more about our expertise and resources, and find out what to expect from longmer screening.
 |
From our solid supports and specialty reagents for oligo modification, to our MerMade™ synthesizers, we provide Our multi-site production capabilities guarantee a resilient supply-chain, with the quality, reliability and scalability you expect. |
References
- Byun J. Recent Progress and Opportunities for Nucleic Acid Aptamers. Life. 2021;11(3):193. doi:10.3390/life11030193
- Yan AC, Levy M. Aptamer-Mediated Delivery and Cell-Targeting Aptamers: Room for Improvement. Nucleic Acid Ther. 2018;28(3):194-199. doi:10.1089/nat.2018.0732
- Nimjee SM, Rusconi CP, Sullenger BA. Aptamers: an emerging class of therapeutics, Annual Review of Medicine 2005;56, 555-583, doi:10.1146/annurev.med.56.062904.144915
- Sun H, et al. Oligonucleotide aptamers: new tools for targeted cancer therapy. Molecular Therapy Nucleic Acids 2014; 3, e182, doi:10.1038/mtna.2014.32
- Ji B, Wei M, Yang B. Recent advances in nanomedicines for photodynamic therapy (PDT)-driven cancer immunotherapy. Theranostics. 2022;12(1):434-458. doi:10.7150/thno.67300
- Photodynamic therapy (PDT). nhs.uk. Published October 19, 2017. Accessed May 1, 2022. https://www.nhs.uk/conditions/photodynamic-therapy/
- Photodynamic Therapy to Treat Cancer - National Cancer Institute. Published September 6, 2011. Accessed May 1, 2022. https://www.cancer.gov/about-cancer/treatment/types/photodynamic-therapy
- Li L, Zhou B, Xu H, Shi H, Gao L, Ge B. Zinc-Loaded Black Phosphorus Multifunctional Nanodelivery System Combined With Photothermal Therapy Have the Potential to Treat Prostate Cancer Patients Infected With COVID-19. Front Endocrinol. 2022;13:872411. doi:10.3389/fendo.2022.872411
- Chen L, Hong W, Duan S, Li Y, Wang J, Zhu J. Graphene quantum dots mediated magnetic chitosan drug delivery nanosystems for targeting synergistic photothermal-chemotherapy of hepatocellular carcinoma. Cancer Biol Ther. 2022;23(1):281-293. doi:10.1080/15384047.2022.2054249
- Ibarra LE, Camorani S, Agnello L, et al. Selective Photo-Assisted Eradication of Triple-Negative Breast Cancer Cells through Aptamer Decoration of Doped Conjugated Polymer Nanoparticles. Pharmaceutics. 2022;14(3):626. doi:10.3390/pharmaceutics14030626
- Lasting impact of lipid nanoparticles. Nat Rev Mater. 2021;6(12):1071-1071. doi:10.1038/s41578-021-00398-6
- Guo L, Shi D, Shang M, et al. Utilizing RNA nanotechnology to construct negatively charged and ultrasound-responsive nanodroplets for targeted delivery of siRNA. Drug Deliv. 2022;29(1):316-327. doi:10.1080/10717544.2022.2026532
- Saify Nabiabad H, Amini M, Demirdas S. Specific delivering of RNAi using Spike’s aptamer‐functionalized lipid nanoparticles for targeting SARS‐CoV‐2: A strong anti‐Covid drug in a clinical case study. Chem Biol Drug Des. Published online 2021. doi:10.1111/cbdd.13978
- Wei J, Song R, Sabbagh A, et al. Cell-directed aptamer therapeutic targeting for cancers including those within the central nervous system. Oncoimmunology. 2022;11(1):2062827. doi:10.1080/2162402X.2022.2062827
- Tsvetkov VB, Varizhuk IV, Kurochkin NN, et al. Anticoagulant Oligonucleotide–Peptide Conjugates: Identification of Thrombin Aptamer Conjugates with Improved Characteristics. Int J Mol Sci. 2022;23(7):3820. doi:10.3390/ijms23073820



