Originally published : Wed, July 6, 2016 @ 5:02 PM
Updated : Wed, March 29, 2023 @ 10:55 AM
Thio C6 Linker (Thiohexyl)
We continue our “Know Your Oligo Mod” series with another versatile custom oligonucleotide modification: the Thio C6 Linker (Thiohexyl) modification. Figure 1 depicts the reactive handle of a Thio C6 Linker that can be incorporated as either a 5’ modification or a 3’ modification on a custom oligonucleotide. The addition of this custom oligo modification creates a conjugation site with a reactive thiol group.
Figure 1:
5' Thio C6 modification:
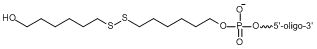
3' Thio C6 modification:

Fig.1 Image shows the chemical structures of the Thio C6 conjugated on the 5’ end (top) or the 3’ end (bottom) of an oligo. Note: this structure represents the protected disulfide form, which is how LGC Biosearch Technologies delivers the oligo. Subsequent reduction of this disulfide bond activates the reactive handle to reveal the free thiol group at the end of a six-carbon linker.
The addition of a thiol group allows the attachment of an oligo to diverse substrates and molecules which include fluorescent dyes, maleimide or halogen compounds, and even the surface of gold nanoparticles. The Thio C6 Linker can also be used to attach an oligonucleotide to peptides or proteins through cysteine residues in the amino acid sequence via disulfide linkages. This is convenient particularly for conjugating oligos to common enzymes such as horseradish peroxidase or alkaline phosphatase.
Clearly, this versatile modification is being used in a continuously expanding number of applications and enables both commonplace and exotic molecular technologies. For example, the strong affinity of thiol groups towards noble metal surfaces has proven effective in producing DNA microarrays containing thiol-modified DNA probes.1 The same covalent bond between thiolated DNA and gold microspheres is also exploited in the fabrication of less common detection methods such as electrochemical biosensors.2 These sensors have been applied in diagnostic applications such as pathogen detection,3,4 disease diagnosis,5 and monitoring transplanted islet cells by magnetic resonance imaging (MRI).6
Thiol-modified oligos are available from our website via the Custom Oligos page. LGC Biosearch Technologies also supplies Thiol C6 CPG and Thiol C6 phosphoramidite for those who prefer to synthesize their own modified oligos.
It is important to note that we supply thiolated oligos in the disulfide form. These oligos must first be activated with a reducing agent prior to their use, as illustrated in Figure 2. The following protocol elaborates on how to reduce the disulfide bond to form free thiol groups:
- Prepare a reaction buffer with 100 mM DTT and 0.2 M sodium phosphate buffer, pH 8.5.
- Bring the oligo into solution with the DTT reaction buffer and incubate at room temperature for 30 minutes to reduce the disulfide bond, leaving an active thiol group.
- Enrich the reduced oligonucleotide product by running the reaction solution through a G-25 nucleic acid purification column.
- Proceed immediately to the next step in conjugation.
Figure 2:
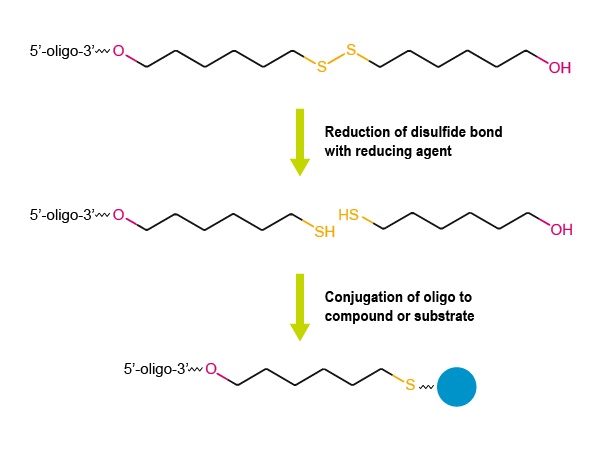
Fig. 2 An oligo modified with a Thio C6 Linker on the 3’ end can be conjugated to other compounds or substrates via the free thiol group (-SH), after reduction of the disulfide bond.
We advise conjugating the thiolated oligo soon after activation because the free thiol groups are reactive. The disulfide linkage will naturally re-oxidize over time between any two thiols in the solution to form a homo-dimer. Therefore, it is imperative to reduce the disulfide bond first if further manipulation of the free thiol is required.
You can explore the Oligo Modifications page at any time to view the wide range of custom oligo modifications we offer. Contact us at techsupport@biosearchtech.com if you are looking for a particular modification not listed on our website. We are frequently able to accommodate requests of special modification to support your oligo needs. Keep an eye out for future posts to the “Know Your Oligo Mod” series or subscribe to never miss another post.
 |
If you are thinking about bringing oligo synthesis in-house, there are many considerations to account for beyond instrument selection. But the benefits of synthesizing your own oligos are clear. Download this guide that summarises all the practical and technical information in a single place, to ensure that your synthesizers run at full capacity and you maximise your return on investment. |
References
- Immobilization techniques for microarray: challenges and applications. Nimse SB, et al. Sensors. 2014 Nov 25;14(12):22208-29. doi: 10.3390/s141222208.
- Electrochemical Aptamer-Based Biosensors: Recent Advances and Perspectives. Abd-Elgawad Radi. Int J Electrochem. 2011 May 16;1-17. doi:10.4061/2011/863196.
- Direct colorimetric diagnosis of pathogen infections by utilizing thiol-labeled PCR primers and unmodified gold nanoparticles. Jung YL, et al. Biosens Bioelectron. 2010 Apr 15;25(8):1941-6. doi: 10.1016/j.bios.2010.01.010.
- A direct detection of Escherichia coli genomic DNA using gold nanoprobes. Padmavathy B, et al. J Nanobiotechnology. 2012 Feb 6;10:8. doi: 10.1186/1477-3155-10-8.
- Colourimetric-based method for the diagnosis of spinal muscular atrophy using gold nanoprobes. Ahmadpour-Yazdi H, et al. IET Nanobiotechnol. 2015 Feb;9(1):5-10. doi: 10.1049/iet-nbt.2013.0065.
- Labeling of islet cells with iron oxide nanoparticles through DNA hybridization for highly sensitive detection by MRI. Kitamura N, et al. Bioorg Med Chem. 2013 Nov 15;21(22):7175-81. doi: 10.1016/j.bmc.2013.08.063.

