Originally published : Thu, December 4, 2014 @ 8:17 PM
Updated : Wed, March 29, 2023 @ 10:56 AM
The chemical synthesis of oligonucleotides allows for the incorporation of a wealth of modifications to an oligo sequence. However, most scientists usually only consider the addition of fluorescent dyes or quenchers to the terminal ends of their oligo to create the prototypical qPCR probe. There exists a wide range of different modification options allowing for a myriad of diverse applications for synthetic nucleic acid chemistry. This article is the first in the Know Your Oligo Mod blog series which will explore the specialty modifications options which Biosearch Technologies makes available for custom oligo synthesis and to highlight some of their potential applications.
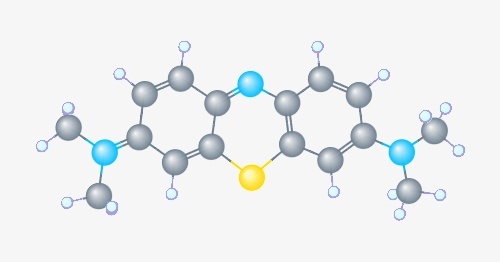
Fig. 1- Chemical Structure of Methylene Blue, Image modified from PubChem record1.
Methylene Blue compound
In this post, we will detail the Methylene Blue modification, which when conjugated to an oligonucleotide can act as an electrochemical biosensor2. The Methylene Blue compound itself has been utilized for over 100 years and has found numerous applications: in medicine as a pharmaceutical treatment3, in pathology as a histological dye4, and in analytical chemistry as a redox indicator5 (Fig. 1). However, more recently, Methylene blue-conjugated oligos have found a use in sensors which take advantage of the compound’s aromatic, heterocyclic structure and redox capabilities. These sensors utilize conformational changes occurring in the oligonucleotide upon interaction with a target molecule, which could be a specific chemical compound, a nucleic acid sequence, or even a protein such as an antibody. These conformational responsive oligos, dubbed aptamers, have been used in a number of other diagnostic applications (an animation of these in action can be found here6). In one example of these biosensors, Methylene Blue modified aptamers are linked through a monolayer to a gold electrode via a thiol linkage (Fig. 2). In this arrangement, the oligo’s conformation (when bound or unbound to its target) controls the level of electron transfer between methylene blue and the electrode. Thus, a measurement of the current running through the electrode provides a means of specific and highly sensitive detection of the target.
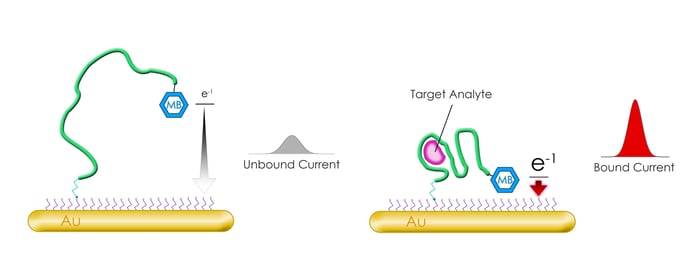
Fig.2 - Example of Methylene Blue labeled oligo utilized as an electrochemical DNA Biosensor. Upon interaction with the target analyte the conformation of the oligo is altered to bring the Methylene Blue in proximity of the electrode affecting the electron transfer (e-1). As a result this system provides an electrical signal readout of the presence of the analyte.
The example above is just one iteration of these novel Methylene Blue electrochemical sensors. Numerous other versions of these sensors have been developed to specifically suit their end use7,8,9. LGC Biosearch Technologies offers four oligo modification options to accommodate various design needs: Methylene Blue can be attached to an oligo either internal to the sequence linked though thymidine base, at the 5’ end of the sequence, or at the 3’ end though a modified thymidine or 7-carbon spacer. Although current means of designing* redox–tagged aptamer biosensors is complex and their results can require the interpretation of an expert, thus far they have shown much promise for sensitive and specific detection of target molecules. As target detection has shown to be successful in various biosensor platforms, including microfluidic devices, it is expected that these electrochemical sensors have a future in numerous fields such as medical diagnostics2,10,11. Biosearch has already successfully manufactured oligos for DNA biosensor assays2,10,12.
Although our specialty lies in oligo design for qPCR and Stellaris RNA FISH, LGC Biosearch Technologies also manufactures custom oligos for use in a broad range of applications and has already successfully manufactured oligos for DNA biosensor assays2,10,12. Methylene Blue labeled oligos can be custom ordered at a variety of scales and purification options, however for use in biosensor development, we recommend dual-HPLC purification. Please contact techsupport@biosearchtech.com for any questions concerning our custom oligo modifications offerings.
* LGC Biosearch Technologies does not currently offer design services of aptamers or specific guidance in their use, rather we provide the custom synthesis and purification of these specialty modified oligos. If interested in additional information or resources about aptamers, please contact our technical support at techsupport@biosearchtech.com
Recommended reads
What's next for aptamers in diagnostics?
Where are all the therapeutic aptamers?
 |
If you are thinking about bringing oligo synthesis in-house, there are many considerations to account for beyond instrument selection. But the benefits of synthesizing your own oligos are clear. Download this guide that summarises all the practical and technical information in a single place, to ensure that your synthesizers run at full capacity and you maximise your return on investment. |
References
- http://pubchem.ncbi.nlm.nih.gov/compound/6099. Accessed Dec. 2, 2014.
- Fabrication of electrochemical-DNA biosensors for the reagentless detection of nucleic acids, proteins and small molecules. Rowe AA, White RJ, Bonham AJ, & Plaxco KW. Journal of Visualized Experiments : JoVE. 2011 June; (52): 1–6. doi:10.3791/2922
- Methylene blue as antidote for cyanide and carbon monoxide poisoning. Brookes MM. J. Am. Med. Assoc. 1933 Jan; 100(1): 59. doi:10.1001/jama.1933.02740010061028.
- Dako Education Guide - Special Stains and H & E second edition Chapter 19: On Chemical Reactions and Staining Mechanisms. Kiernan JA.
- Visualizing Redox Chemistry: Probing Environmental Oxidation–Reduction Reactions with Indicator Dyes. Tratnyek PG, et al. Chem. Educ. 2001 June; 6(3): 172–179. doi:10.1007/s00897010471a
- Electrochemical Aptamer-Based Biosensors: Recent Advances and Perspectives. Radi A-E. International Journal of Electrochemistry. 2011 May; 1–17. doi:10.4061/2011/863196
- Electrochemical Behaviors of Methylene Blue on DNA Modified Electrode and Its Application to the Detection of PCR Product from NOS Sequence. Zhu L, Zhao R, Wang K, Xiang H, et al. Sensors. 2008 Sept; 8(9), 5649–5660. doi:10.3390/s8095649
- An Electrochemical Supersandwich Assay for Sensitive and Selective DNA Detection in Complex Matrices. Xia F, White RJ, Zuo X, et al. .J Am Chem Soc. 2010 Oct; 132(41): 14346–14348. doi:10.1021/ja104998m
- Aptamer-based Electrochemical Biosensor for Interferon Gamma Detection. Liu Y, Tuleouva N, Ramanculov E, & Revzin A. Anal Chem. 2010 Oct; 82(19): 8131–8136. doi:10.1021/ac101409t.
- Continuous, Real-Time Monitoring of Cocaine in Undiluted Blood Serum via a Microfluidic, Electrochemical Aptamer-Based Sensor. Swensen JS, et al. J. Am. Chem. Soc. 2009; 131: 4262–4266. doi:10.1021/ja806531z
- MEDIC to kick-start personalised medicine revolution. Gross M. Published: November 27, 2013. Accessed: December 3, 2014. http://www.rsc.org/chemistryworld/2013/11/medic-sensor-kick-start-personalised-medicine-revolution
- Optimization of Electrochemical Aptamer-Based Sensors via Optimization of Probe Packing Density and Surface Chemistry. White RJ, Phares N, Lubin AA, Xiao Y, & Plaxco KW. Langmuir. 2008 Sept; 24(18): 10513–10518. doi:10.1021/la800801v.

