Originally published : Thu, July 23, 2015 @ 5:33 PM
Updated : Fri, January 13, 2023 @ 11:35 AM
In this instalment, we will cover the basics of LGC Biosearch Technologies’ line of proprietary non-fluorescent quencher chromophores for oligonucleotide modification. The Black Hole Quencher ™ (BHQ™) dyes are often abbreviated to just BHQ dyes and are numbered according to the range of the visible spectrum in which they are most effective, from zero to three. Before exploring all the different BHQ dye modifications, we should explain a bit about what a quencher actually is and how they are used as oligonucleotide modifications.
FRET quenching
Quenchers, in general, are a class of molecules with the capability to suppress the fluorescence emission intensity via a mechanism known as fluorescence resonance energy transfer, or simply FRET. In brief, this process requires the emission spectrum of the reporter dye (donor) to have sufficient spectral overlap with the absorbance spectrum quencher dye (acceptor). FRET also requires the two molecules to be in the correct relative orientation while maintaining within close spatial proximity. When these conditions are met, the FRET mechanism’s transfer of energy from the excited reporter dye to the quencher returns the reporter dye to its ground state in a non-radiative means without any light emission or absorption between the two dyes. Simultaneously, via a dipole coupling, the absorbed energy of the acceptor raises the quencher to an excited state. The quencher will then dissipate this energy proportional to the transferred energy from the donor dye. Should the quencher be another fluorophore such as tetramethylrhodamine (TAMRA), this would typically present light emission from the acceptor dye. The BHQ dyes are further differentiated in their quenching capabilities because they are members of a special class of quencher dyes known as “Dark Quenchers”. These dyes have broad absorption ranges in the visible spectrum, but have no native fluorescence themselves, in contrast to non-dark quenchers like TAMRA, which can contribute to background signal in an assay. This is part of the secret to the four Black Hole Quencher dyes’ ability to suppress signal from a wide range of fluorophores without potentially contributing to background fluorescence, enabling highly multiplexed nucleic acid assays.
The FRET quenching mechanism relies on spatial proximity and overlap of the spectral characteristics of the reporter’s emission and the quencher’s absorbance. When in close proximity, typically within 10 nanometers distance to one another, there is an electronic interaction in which a transfer of energy from the reporter dye to the quencher results in the transferred energy dissipating via the quencher. If the quencher is a dark quencher such as BHQ, the energy does not cause photon emission but rather is partially released in the form of heat from the quencher as it relaxes to its ground state. To put more simply, BHQ quenching via the FRET mechanism takes the excited reporter dye’s energy and transforms it so that the reporter dye returns to a ground state while suppressing any release of light.
Static quenching
While BHQ dyes participate in the more well know FRET Quenching mechanism, they can also efficiently operate via Static (contact) quenching mechanisms, elucidated in a series of studies from 2002 1-3. The principles behind both of these quenching mechanisms are quite complex but are clearly explained in detail on the following webpage and the references cited below. The second mechanism of quenching is referred to as contact quenching, also known as static quenching. Through this mechanism, the fluorophore and quencher physically interact to form an intramolecular dimer. This dimerisation of the two chromophores essentially creates a new, non-fluorescent compound. Static quenching relies on the affinity of the dye and the quencher for one another. Because BHQ dyes and many fluorophores are planar hydrophobic molecules, they will often strongly associate in aqueous solutions, thus assisting in creating these intramolecular dimers. In the case of either quenching mechanism, there is a requirement to achieve a high local concentration of both the fluorophore and quencher. As such, quenching mechanisms are typically not effective as free chromophore monomers, but rather the reporter dye and quencher must be closely tethered to each other by linking them to the same molecule, as is the case in a Dual-Labeled BHQ probe. Black Hole Quencher dyes have the capacity to operate through both mechanisms of quenching, allowing BHQ-conjugated oligos to effectively snuff out the signal from reporter dyes.
Selecting the appropriate non-fluorescent quencher
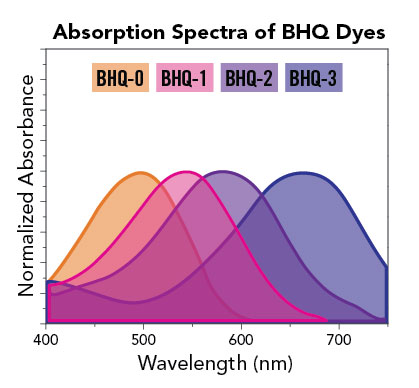
One important consideration in choosing the correct BHQ dye is to ensure you select a quencher with an absorption range best matched to the fluorescent reporter. There are some additional recommendations for dye-quencher pairings, which are particularly suited to static quenching mechanisms, such as partnering our Quasar® 670 and Quasar 705 dyes with the BHQ-2 quencher, even though the BHQ-3 quencher would appear to provide the optimal spectral overlap. Therefore, certain dyes may have multiple, effective quencher options available. If you are ever unsure about which non- fluorescent quencher to choose, we offer handy tools to help make these decisions in our Dye Selection Chart and Spectral Overlay Tool, or simply ask our technical services team at techsupport@biosearchtech.com.
Available Black Hole Quencher modifications for custom oligonucleotides
| Dye | Effective quenching range |
5’ Mod. | 3’ Mod. | Internal mod. |
| BHQ-0 | 430-520 nm | ✔ | ✔ | |
| BHQ-1 | 480-580 nm | ✔ | ✔ | ✔ |
| T(BHQ-1) | 480-580 nm | ✔ | ✔ | |
| BHQ-2 | 559-670 nm | ✔ | ✔ | ✔ |
| T(BHQ-2) | 559-670 nm | ✔ | ✔ | |
| BHQ-3 | 620-730 nm | ✔ | ✔ | ✔ |
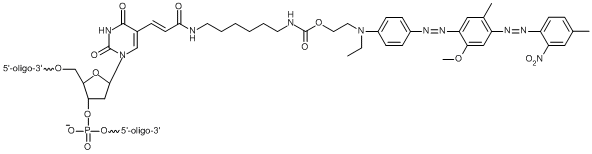
BHQ modifications to oligos can be used in a number of common probe formats for molecular assays, including qPCR, such as Dual-Labeled BHQ probes and BHQplus® probes. These modifications are also used in a number of more exotic arrangements available through our custom oligo synthesis offering. To accommodate such a broad range of uses and oligo constructs, LGC Biosearch offers multiple versions of our BHQ dye modification to allow different positioning of the quencher (see table above). All BHQ dyes are available as terminal 5’ and 3’ modifications to an oligonucleotide. Many of them are also available as internal BHQ modifications, which come in two versions. Standard internal BHQ modifications incorporate an abasic linkage through the sugar-phosphate backbone of the oligo. This version of the internal BHQ may impact oligonucleotide geometry upon hybridization due to the fact that it sits between the standard base linkages on either side of the modification. There is also a version of the internal modification, T(BHQ), where the quencher is attached to a deoxythymidine (dT) nucleoside to allow continuity of the oligonucleotide backbone. This minimizes the potential disruption of hybridization by linking the quencher off the base. The structure of the BHQ-1 abasic and T-linked modifications are shown side by side here as point of comparison.
Internal BHQ-1 Modification // [BHQ-1]
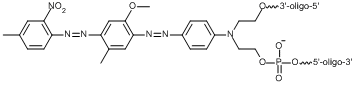
Internal Thymidine linked BHQ-1 Modification // [T(BHQ-1)]
The number of available Black Hole Quencher dyes and modification arrangements presents a large degree of customization options for your oligos. Static and FRET quenching mechanisms of the BHQ line of dyes provides effective quenching across the visible spectrum. To find out more about the BHQ dyes and the history of quencher evolution, check out our quenching mechanisms applications page and qPCR Design Lab’s qPCR & BHQ page.
 |
If you are thinking about bringing oligo synthesis in-house, there are many considerations to account for beyond instrument selection. But the benefits of synthesizing your own oligos are clear. Download this guide that summarises all the practical and technical information in a single place, to ensure that your synthesizers run at full capacity and that you maximise your return on investment. |
References
- Efficiencies of fluorescence resonance energy transfer and contact-mediated quenching in oligonucleotide probes.Salvatore A. E. Marras, Fred Russell Kramer and Sanjay Tyagi. Nucleic Acids Research. 2002. 30. e122.
- Intramolecular Dimers: A New Strategy to Fluorescence Quenching in Dual-Labeled Oligonucleotide Probes.Mary Katherine Johansson, Henk Fidder, Daren Dick and Ronald M. Cook. J. Am. Chem. Soc. 2002, 124, 6950.
- Choosing Reporter-Quencher Pairs for Efficient Quenching Through Formation of Intramolecular Dimers. Mary Katherine Johansson. Methods Mol Biol. 2006, 335, 17.

