Originally published : Tue, February 23, 2010 @ 7:19 PM
Updated : Mon, September 19, 2022 @ 2:10 PM
A BHQ® probe is a dual-labeled oligonucleotide covalently labeled with a fluorophore and a Black Hole Quencher® (BHQ) dye.
Structure
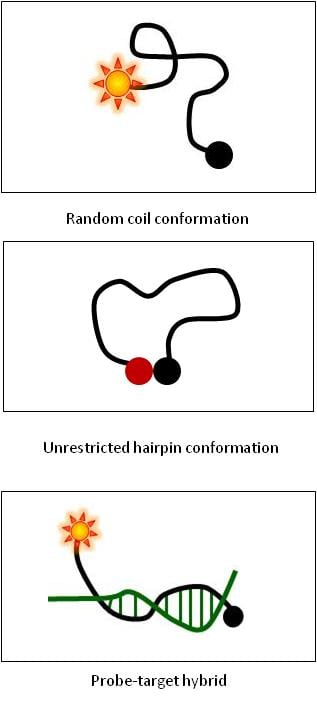
Q. What are the main components of a dual-labeled Black Hole Quencher® (BHQ) probe?
- An oligonucleotide, typically 30 bases long
- A 3' BHQ dye
- A 5'-fluorophore (reporter) dye
Q. What is its native conformation at melting and annealing temperatures?
Melted - random coil conformation
Unhybridized - unrestricted hairpin, FRET-quenched (no signal)
Hybridized- stable double helix, probe-target hybrid (signal)
Mechanism
Q. How does it yield signal?
When a complementary sequence is available, the probe hybridizes to the complementary sequence. Conformational changes associated with hybridization separate the fluorophore and quencher, decreasing FRET quenching and releasing fluorescence.
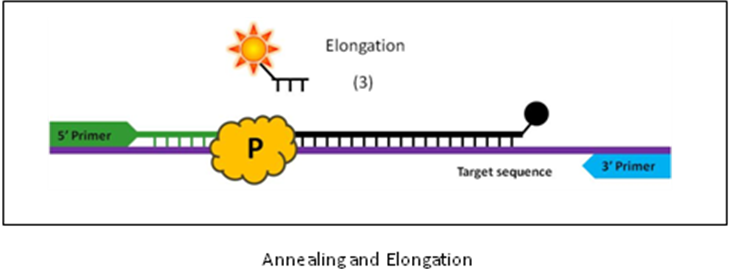
Q. What happens with each consecutive PCR cycle?
1. Heat melts or denatures the probe, sense and antisense strands of a DNA duplex.
2. As temperatures cool, hydrophobicity and electrostatics promote dye-dye attractions and enhance fluorescent quenching.
3. At annealing temperatures, the primers and the BHQ probe anneal to their complementary sequences within the target DNA. Conformational changes during hybridization separate the dyes which decreases FRET quenching thus releasing fluorescence.
4. During elongation, the DNA polymerase incorporates nucleotides complementary to the strand as it progresses in a 5' to 3' direction from the primer. When the polymerase encounters the 5'-end of the probe, it cleaves off the nucleotide, or a flap of nucleotides, with the bound reporter dye, thereby permanently separating the reporter and quencher dyes.
.
Advantages of Dual-labeled BHQ probes
Dual-labeled BHQ probes have replaced earlier reporter-quencher dye pairings, such as FAM-TAMRA or FAM-DABCYL. In such sub-optimal probes, the quencher has inherent limitations such as auto-fluorescence or insufficient quenching at certain wavelengths which limit the choice of quenchable fluorophores. In contrast, the BHQ dyes:
- are highly efficient dark quenchers;
- have broad absorption spectra;
- and yield high signal to noise ratios.
BHQ dyes can be paired with all common reporter dyes emitting between the ultraviolet and infrared wavelengths, thereby making multiplexed hybridisation assays easy to design and interpret.
Applications: Single and multiplex, quantitative and qualitative, real-time and endpoint PCR analyses; allelic discrimination; and SNP detection.
Written by: Christina Ferrell, Ph.D., Technical Applications Specialist
- Didenko, V.V. "DNA probes using Fluorescence Resonance Energy Transfer (FRET): Designs and Applications". BioTechniques 31 (2001): 1106-1121. (Review)
- Bustin, S.A. A-Z of Quantitative PCR. (IUL Biotechnology Series). La Jolla, California: International University Line, 2004.
- Ranasinghe, R.T., Brown, T. "Fluorescence based strategies for genetic analysis". Chem. Commun. (2005): 5487-5502.
- Johansson, M.K. "Choosing Reporter-Quencher Pairs for Efficient Quenching Through Formation of Intramolecular Dimers." Methods in Molecular Biology (v. 335, ch. 2). Ed. V.V. Didenko. Totowa, NJ: Humana Press, 2004. 17-29.
- Marras, S.A.E. "Fluorescent Energy Transfer Nucleic Acid Probes: Designs and Protocols" Methods in Molecular Biology (v. 335, ch. 7). Ed. V.V. Didenko. Totowa, NJ: Humana Press, 2004. 3-16.
- Biosearch Technologies website. Black Hole Quencher Dyes
- Biosearch Technologies website. Genotyping-qPCR

